SQF Readiness
Map clauses to documents, evidence, tasks, findings, and responsible team members.
Certiflow HQ is a web-based food safety compliance platform designed to help companies centralize SQF readiness, document control, audits, training, recipes, inventory, traceability, and automated reporting.
Certiflow HQ brings the moving parts of SQF readiness and daily food safety operations into one structured workspace, so policies, procedures, records, training, reports, and evidence are not scattered across binders, spreadsheets, and mystery folders.
Map clauses to documents, evidence, tasks, findings, and responsible team members.
Manage SOPs, versions, approvals, review dates, obsolete files, and acknowledgments.
Run internal audits, track findings, assign corrective actions, verify closure, and preserve proof.
Assign role-based training, track completion, maintain records, and monitor retraining due dates.

Certiflow HQ is designed for food businesses, quality teams, food safety managers, SQF practitioners, consultants, and multi-site operators who need consistent records, clear accountability, and audit-ready proof.

Food safety programs fail quietly before they fail loudly. Missing records, old SOP versions, incomplete training, open corrective actions, and disconnected logs pile up until audit week turns into a fire drill wearing a hairnet.
Keep records, documents, evidence, signoffs, and reports connected to the requirements they support.
See gaps earlier with dashboards, due dates, reminders, missing evidence alerts, and readiness views.
Support proprietary recipes, procedures, suppliers, and customer data with separated tenant workspaces.

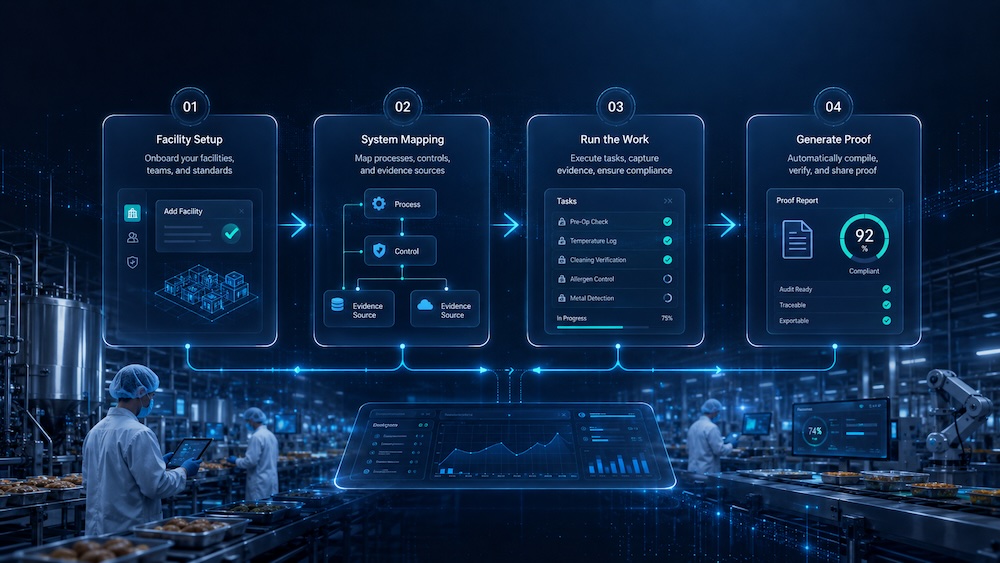
Certiflow HQ is being designed to guide each facility through setup, evidence collection, daily execution, corrective action tracking, and report generation from one command center.
Define company details, users, roles, facilities, modules, SQF scope, and starter templates.
Connect requirements to SOPs, records, training, suppliers, recipes, logs, and operational evidence.
Assign tasks, conduct audits, close findings, approve documents, and monitor training status.
Create readiness summaries, management review packets, audit reports, and traceability documentation.
Start with SQF readiness and document control, then expand into training, recipes, ingredient conversions, inventory, supplier management, traceability, and automated reporting as the business grows.

Certiflow HQ is a web-based SaaS platform at certiflowhq.com, built to serve client workspaces through secure company subdomains and scalable compliance modules.
